- Latest news▼
-
15:11, April 24 Zombie deer disease possibly linked to hunters’ deaths

-
12:27, April 23 Appetite: Scientists found out the secret to the appeal of large portions of fast food

-
10:33, April 23 Scientists test new approach to fighting viruses

-
08:38, April 23 Ketamine may help with postpartum depression

-
22:12, April 22 Unhealthy amount of sugar found in baby food products of a well-known brand

-
19:41, April 22 Air pollution puts health of more than 1.6 billion workers globally at risk

-
17:25, April 22 Scientists found baked goods and lack of sleep to be more dangerous than alcohol

-
16:02, April 22 342 cases of measles recorded in Armenia so far in 2024
-
15:29, April 22 BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly

-
08:27, April 22 Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds

-
20:37, April 21 Environmental Health Perspectives: Microplastics ingested with food and water can spread from the gut to the brain

-
22:41, April 20 Scientists develop new method to safely stimulate immune cells to fight cancer

-
20:46, April 20 Blood test can determine who is at risk of developing multiple sclerosis - scientists

-
18:36, April 20 Next pandemic likely to be triggered by flu - scientists

-
12:16, April 19 Scientists grow human mini-lungs in lab

All materials
Lab-grown bone cell breakthrough heralds new benefits for orthopaedics
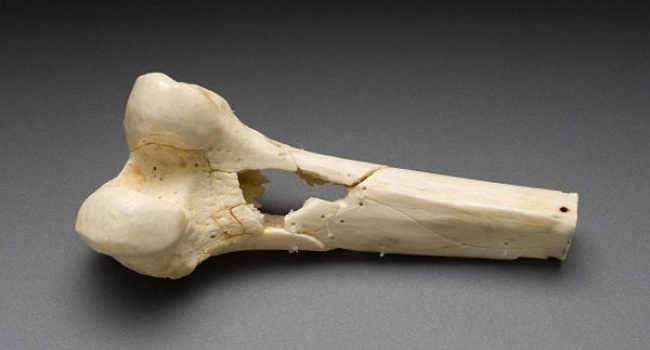
Technology originally developed to detect gravitational waves is being used to generate tissue engineered bone grafts for future use in orthopaedic medicine, scientists report in a new paper published today.
The latest development in a technique known as 'nanokicking' has allowed scientists from the Universities of Glasgow, Strathclyde, the West of Scotland and Galway to grow three-dimensional samples of mineralised bone in the laboratory for the first time.
Bone is the second most grafted tissue after blood and is used in reconstructive, maxillofacial and orthopaedic surgeries. Currently, however, surgeons can only harvest limited amounts of living bone from the patient for use in graft, and bone from other donors is likely to be rejected by the body. Instead, surgeons must rely on inferior donor sources which contain no cells capable of regenerating bone, limiting the size of repairs they can effect.
In a paper in the journal Nature Biomedical Engineering, the researchers describe how they have used measurement technology, based on the sophisticated laser interferometer systems built for gravitational wave detection of astrophysical objects, to turn mesenchymal cells taken from human donors into bone cells in three dimensions. These 3D living bone grafts, when implanted into patients in the future, will be able to repair or replace damaged sections of bone.
Mesenchymal stem cells, which are naturally produced by the human body in bone marrow, have the potential to differentiate into a range of specialised cell types such as bone, cartilage, ligament, tendon and muscle. Nanokicking subjects cells to ultra-precise, nanoscale vibrations while they are suspended inside collagen gels. The process of nanokicking turns the cells in the gels into a 'bone putty' that has potential to be used to heal bone fractures and fill bone where there is a gap. Using patients' own mesenchymal cells means surgeons will be able to prevent the problem of rejection, and can bridge larger gaps in bone.
Matthew Dalby, professor of cell engineering at the University of Glasgow, is one of the lead authors of the paper.
Professor Dalby said: "This is an exciting step forward for nanokicking, and it takes us one step further towards making the technique available for use in medical therapies. We are especially excited by these developments as much of the work we're doing now is funded by Sir Bobby Charlton's landmine charity Find a Better Way, which help individuals and communities heal from the devastating impact of landmines and other explosive remnants of war.
"Now that we have advanced the process to the point where it's readily reproducible and affordable, we will begin our first human trials around three years from now in the NHS along with the Scottish National Blood Transfusion Service and reconstructive and orthopaedic surgeons in Glasgow."
Find A Better Way CEO Lou McGrath said: "Producing synthetic, off-the-shelf bone tissue will potentially transform the lives of untold numbers of civilian landmine blast survivors around the world. Find A Better Way is delighted to be funding this project - it is a perfect match for Sir Bobby's dream of devising new solutions for one of the world's most intractable set of problems."
The Find A Better Way project at the University of Glasgow is led by professor of bioengineering Manuel Salmeron-Sanchez. In the Find a Better Way project, the team will combine the bone putty with large 3D printed scaffolds to fill even larger bone defects.
Prof Salmeron-Sanchez recently visited Cambodia to meet local people who have suffered landmine-related injuries. He added: "For many people who have lost legs in landmine accidents, the difference between being confined to a wheelchair and being able to use a prosthesis could be only a few centimetres of bone".
Professor Dalby said: "In partnership with Find A Better Way, we have already proven the effectiveness of our scaffolds in veterinary medicine, by helping to grow new bone to save the leg of a dog who would otherwise have had to have it amputated. Combining bone putty and mechanically strong scaffolds will allow us to address large bone deficits in humans in the future."
Some of the technology which underpins the nanokicking technique was originally developed by astrophysicists working on the search for gravitational waves, ripples in spacetime caused by massive events such as the collision of black holes.
Stuart Reid, Professor of Biomedical Engineering at the University of Strathclyde, and formerly at the University of the West of Scotland, said: "Having spent 15 years working in astrophysics and gravitational wave detectionwith the Laser Interferometer Gravitational-Wave Observatory (LIGO), it is amazing to see technology arising that could revolutionise key aspects of tissue engineering and regenerative medicine. The team is hard at work to get the technology ready for the first human trials, and to get devices into other labs around the UK and further afield."
The nanokick bioreactors developed by the researchers are currently being further tested in a network of laboratories across the UK. Since mesenchymal cells have the potential to differentiate into numerous other types of cell in addition to bone, the researchers expect that other clinically relevant applications of nanokicking will be discovered in partner labs in the future.
The team's paper, titled 'Stimulation of 3D osteogenesis by mesenchymal stem cells using a nanovibrational bioreactor', is published in Nature Biomedical Engineering.
Follow NEWS.am Medicine on Facebook and Twitter
- Video
- Event calendar
- Archive
- Most read
month
week
day
- JAMA Oncology: Urine test can help rule out high-grade prostate cancer with almost 100% accuracy, study shows 1216
- Daily Mail: Elderly woman in China gets infected with brain-eating amoeba 1168
- The Conversation: childhood trauma can cause pathological hoarding 1152
- Obesity: exercising before breakfast helps you lose weight faster 1152
- Daily Mail: Satiating food reduces cravings for sweets, nutritionist says 1126
- Scientists grow human mini-lungs in lab 1117
- Next pandemic likely to be triggered by flu - scientists 721
- Scientists found baked goods and lack of sleep to be more dangerous than alcohol 646
- 342 cases of measles recorded in Armenia so far in 2024 608
- Blood test can determine who is at risk of developing multiple sclerosis - scientists 597
- Scientists develop new method to safely stimulate immune cells to fight cancer 590
- Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds 573
- BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly 530
- Air pollution puts health of more than 1.6 billion workers globally at risk 366
- Ketamine may help with postpartum depression 357
- Find us on Facebook
- Poll