- Latest news▼
-
19:41, April 25 Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference

-
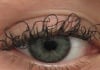
17:31, April 25 JAMA: patient grew long, curly eyelashes because of chemotherapy
-
11:08, April 25 Mpox epidemic declared in Republic of the Congo

-
08:31, April 25 OU: quitting smoking 4 times more likely to cure laryngeal cancer

-
01:20, April 25 Paralyzed man in China writes hieroglyphs using neural implants placed in his brain

-
15:11, April 24 Zombie deer disease possibly linked to hunters’ deaths

-
12:27, April 23 Appetite: Scientists found out the secret to the appeal of large portions of fast food

-
10:33, April 23 Scientists test new approach to fighting viruses

-
08:38, April 23 Ketamine may help with postpartum depression

-
22:12, April 22 Unhealthy amount of sugar found in baby food products of a well-known brand

-
19:41, April 22 Air pollution puts health of more than 1.6 billion workers globally at risk

-
17:25, April 22 Scientists found baked goods and lack of sleep to be more dangerous than alcohol

-
16:02, April 22 342 cases of measles recorded in Armenia so far in 2024
-
15:29, April 22 BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly

-
08:27, April 22 Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds

All materials
Can scientists rejuvenate an aging brain with a protein found in umbilical cord blood?

What old brains need is a shot of young blood — and the younger the better.
It may sound like a metaphor employed by a randy octogenarian. But new research on mice suggests it can be taken quite literally.
Writing in the journal Nature, Stanford University anti-aging researchers reported Wednesday that they have found a “restorative factor for the aged hippocampus.” It’s a protein called tissue inhibitor of metalloproteinases 2, or TIMP2, that is found in the blood of young humans — and most copiously in umbilical cord blood.
Harvested at the time of an infant’s birth, cord blood is a rich source of many known regenerative substances, including hematopoietic stem cells, the kind found in bone marrow. Cord blood is capable of treating more than 80 diseases, including blood cancers, inherited blood diseases such as sickle cell and thalassemia, and a range of immune deficiencies.
What makes a grocery store great? Well-labeled, quality produce with a better, more convenient shopper experience. Here’s our list of America’s best grocery stores to serve you in 2017.
In mice that were roughly the equivalent of 70 human years old, three injections of human cord plasma woke up a host of dormant genes in the brain. The injections also increased the flagging snap, crackle and pop of synapses in the hippocampus — a brain region that's crucial to memory.
In addition, the cord blood improved the performance of aged mice as they engaged in memory and learning tasks, such as maze-running and fear-conditioning exercises.
Mice who got injections of blood from humans with a median age of 22 saw some but not all of these improvements, and they were more modest. But the unlucky mice that got blood from healthy “elderly” human subjects (with a median age of 66 years) saw no improvement at all.
The new study draws on a decades-old line of research called parabiosis, in which scientists lashed together the circulatory systems of young and old animals in a bid to mingle their blood and improve age-related decline in the older animal.
In a pathbreaking 2014 study, a team led by the senior author of the latest research, Stanford regenerative scientist Tony Wyss-Coray, showed that many of the beneficial effects seen from parabiosis experiments — including strengthened heart and other muscle tissue — could be had with repeated injections of plasma from young animals.
For the new study, Wyss-Coray’s team aimed to zero in on the precise components of blood that return an older body to youthful function — and to look for those so-called “factors” in human plasma. They focused on the hippocampus, a structure in the brain whose performance clearly suffers with age. Indeed, it is also one of the first points of attack when the brain comes under assault by diseases of age, such as Alzheimer’s.
An ordinary lab mouse would mount a massive immune response to the introduction of human blood in its veins, with certain fatal result. So to begin their explorations, Wyss-Coray’s team used mice bred to be immunodeficient. Then, after identifying proteins with potential regenerative powers, the researchers isolated them and tested them in ordinary, healthy mice.
So, what factor in the human cord blood was awakening the tired hippocampi of elderly mice? Solving thatmystery required an exhaustive process of documenting, measuring and comparing concentrations of several proteins in the blood of mice and humans at different ages. Ultimately, the researchers began to observe that TIMP2 was especially plentiful in cord blood, but declined precipitously with age.
And when they saw that treatment with TIMP2 energized production of certain cells at the heart of the hippocampus, the researchers suspected they had their regenerative elixir. When they attached chemical tracers to the TIMP2 protein and injected it into the elderly mice, they could see it take up residence in the hippocampus, and saw that it “robustly increased” the electrical signals by which cells there communicate. And the older mice demonstrated improvements in learning and memory tasks.
Those behavioral improvements are hard to quantify, but amounted to a boost ranging from 30% to 50%, Wyss-Coray said.
“They don’t function like a young mouse,” he said. “But they maybe get halfway in that direction.”
Their improvements were especially evident in tasks involving spatial memory, an aspect of memory that suffers early in Alzheimer’s disease.
TIMP2 appears to have “quite an important function,” Wyss-Coray said, and “could almost be a master regulator” of processes that are key to aging. Other researchers, he noted, have explored its role in inhibiting the growth and spread of cancer, another disease of aging.
Finding that TIMP2 can alter the activity of the hippocampus as well as complex behavior in mice is a far cry from showing it can be an effective agent of neural regeneration in humans, Wyss-Coray said. But it’s an important waystation on the path to showing its potential in humans. The process of producing it in recombinant form, purifying it as a treatment, and testing it extensively in humans could take five to 10 years, he added.
Wyss-Coray co-directs Stanford’s Alzheimer’s Disease Research Center and is associate director of the Center for Tissue Regeneration, Repair and Restoration at the Palo Alto (Calif.) Veterans Affairs Health Service. He co-founded a company, Alkahest, that is conducting an early clinical trial in which plasma from young donors is intravenously administered to 18 people with mild to moderate Alzheimer's disease. That trial is complete and the results are expected soon.
Follow NEWS.am Medicine on Facebook and Twitter
- Video
- Event calendar
- Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference
- First Armenian-German Conference entitled “Heart Failure Spring School”
- Allogeneic bone marrow transplant in case of hematological malignancy performed in Armenia for first time
All materials
- Archive
- Most read
month
week
day
- Scientists found baked goods and lack of sleep to be more dangerous than alcohol 976
- Next pandemic likely to be triggered by flu - scientists 973
- 342 cases of measles recorded in Armenia so far in 2024 929
- Scientists develop new method to safely stimulate immune cells to fight cancer 794
- Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds 791
- Blood test can determine who is at risk of developing multiple sclerosis - scientists 787
- BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly 728
- Unhealthy amount of sugar found in baby food products of a well-known brand 708
- Ketamine may help with postpartum depression 708
- Appetite: Scientists found out the secret to the appeal of large portions of fast food 699
- Air pollution puts health of more than 1.6 billion workers globally at risk 698
- Scientists test new approach to fighting viruses 685
- Zombie deer disease possibly linked to hunters’ deaths 606
- Mpox epidemic declared in Republic of the Congo 401
- Paralyzed man in China writes hieroglyphs using neural implants placed in his brain 360
- Find us on Facebook
- Poll