- Latest news▼
-
15:32, April 26 ESCMID: New method of purifying the air with ultraviolet light could protect world from new pandemic

-
08:43, April 26 Enzymes that convert different blood groups into first group are discovered

-
19:41, April 25 Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference

-
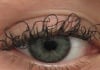
17:31, April 25 JAMA: patient grew long, curly eyelashes because of chemotherapy
-
11:08, April 25 Mpox epidemic declared in Republic of the Congo

-
08:31, April 25 OU: quitting smoking 4 times more likely to cure laryngeal cancer

-
01:20, April 25 Paralyzed man in China writes hieroglyphs using neural implants placed in his brain

-
15:11, April 24 Zombie deer disease possibly linked to hunters’ deaths

-
12:27, April 23 Appetite: Scientists found out the secret to the appeal of large portions of fast food

-
10:33, April 23 Scientists test new approach to fighting viruses

-
08:38, April 23 Ketamine may help with postpartum depression

-
22:12, April 22 Unhealthy amount of sugar found in baby food products of a well-known brand

-
19:41, April 22 Air pollution puts health of more than 1.6 billion workers globally at risk

-
17:25, April 22 Scientists found baked goods and lack of sleep to be more dangerous than alcohol

-
16:02, April 22 342 cases of measles recorded in Armenia so far in 2024
All materials
Alzheimer's drug trial in U.S. results in death of patient

A 65-year-old American woman died of a massive brain hemorrhage during an experimental treatment with a drug called lecanemab for Alzheimer's disease. The drug lecanemab, which is an antibody, is being developed by Biogen in collaboration with the Japanese pharmaceutical company Eisai.
According to Science, the woman received injections as part of a clinical trial and suffered a stroke and bleeding that had previously been seen with similar antibodies. After the stroke in the emergency room at Northwestern University Medical Center in Chicago, she was prescribed the drug to break up the blood clots. However, she could not be saved; the woman died a few days later.
In September, Eisai and Biogen reported positive results from a phase III clinical trial of lekanemab. According to the results, after 18 months from the beginning of the study among volunteers with moderate cognitive impairment and early stage dementia, the drug showed a 27% reduction in cognitive impairment compared to the control group.
Biogen shares fell 5.35% to $288.88 a share in premarket trading on Nov. 28 following the news of the patient's death, NASDAQ trading data showed. Biogen's stock rebounded after the U.S. market opened the session and was trading at $299.19 a share as of 5:41 p.m. Moscow time.
Follow NEWS.am Medicine on Facebook and Twitter
- Video
- Event calendar
- Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference
- First Armenian-German Conference entitled “Heart Failure Spring School”
- Allogeneic bone marrow transplant in case of hematological malignancy performed in Armenia for first time
All materials
- Archive
- Most read
month
week
day
- Scientists found baked goods and lack of sleep to be more dangerous than alcohol 1041
- Next pandemic likely to be triggered by flu - scientists 1017
- 342 cases of measles recorded in Armenia so far in 2024 965
- Unhealthy amount of sugar found in baby food products of a well-known brand 831
- Air pollution puts health of more than 1.6 billion workers globally at risk 827
- Scientists develop new method to safely stimulate immune cells to fight cancer 825
- Ketamine may help with postpartum depression 824
- Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds 824
- Blood test can determine who is at risk of developing multiple sclerosis - scientists 822
- Appetite: Scientists found out the secret to the appeal of large portions of fast food 818
- Scientists test new approach to fighting viruses 799
- BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly 754
- Zombie deer disease possibly linked to hunters’ deaths 744
- Mpox epidemic declared in Republic of the Congo 552
- Paralyzed man in China writes hieroglyphs using neural implants placed in his brain 523
- Find us on Facebook
- Poll