- Latest news▼
-
19:41, April 25 Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference

-
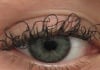
17:31, April 25 JAMA: patient grew long, curly eyelashes because of chemotherapy
-
11:08, April 25 Mpox epidemic declared in Republic of the Congo

-
08:31, April 25 OU: quitting smoking 4 times more likely to cure laryngeal cancer

-
01:20, April 25 Paralyzed man in China writes hieroglyphs using neural implants placed in his brain

-
15:11, April 24 Zombie deer disease possibly linked to hunters’ deaths

-
12:27, April 23 Appetite: Scientists found out the secret to the appeal of large portions of fast food

-
10:33, April 23 Scientists test new approach to fighting viruses

-
08:38, April 23 Ketamine may help with postpartum depression

-
22:12, April 22 Unhealthy amount of sugar found in baby food products of a well-known brand

-
19:41, April 22 Air pollution puts health of more than 1.6 billion workers globally at risk

-
17:25, April 22 Scientists found baked goods and lack of sleep to be more dangerous than alcohol

-
16:02, April 22 342 cases of measles recorded in Armenia so far in 2024
-
15:29, April 22 BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly

-
08:27, April 22 Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds

All materials
Chinese authorities approve drug for Alzheimer's

Chinese authorities have approved a drug for the treatment of Alzheimer's disease - the first new drug with the potential for the treatment of cognitive disorders in 17 years, CNN reported.
According to a statement by the Chinese Drug Safety Agency, a seaweed-based drug called Oligomannate can be used to treat mild to moderate Alzheimer's disease. However, this statement is conditional, which means that although it may go on sale during additional clinical trials, it will be strictly monitored and may be withdrawn in the event of any problems.
In September, a new drug development team led by Geng Meiyu of theShanghai Institute of Materia Medica Institute under the Chinese Academy of Sciences said they were inspired by the study of algae because of the relatively low incidence of Alzheimer's among people who regularly consume it.
In an article in the Cell Research, the Geng team described how sugar contained in algae suppresses some bacteria found in the intestines that can cause nervous degeneration and inflammation of the brain, leading to Alzheimer's disease.
This mechanism was confirmed in a clinical trial conducted by Green Valley, a Shanghai-based pharmaceutical company that will launch the new drug on the market.
"Conducted on 818 patients, the trial found that Oligomannate -- which is derived from brown algae -- can statistically improve cognitive function among people with Alzheimer's in as little as four weeks, according to a statement from Green Valley," the source noted.
Vincent Mok, who heads the neurology division at the Chinese University of Hong Kong, said the new drug showed "encouraging results" when compared to acetylcholinesterase inhibitors - the existing treatment for mild to severe Alzheimer's.
"It is just as effective but it has fewer side effects," he told CNN. "It will also open up new avenues for Alzheimer's research, focusing on the gut microbiome."
Since very little is known about the mechanisms of the new drug, Mok said it should also be checked whether it can have a protective effect and, possibly, slow the progression of the disease in patients who have not yet developed severe symptoms of dementia.
"The company said Oligomannate will be available in China "very soon," and it is currently seeking approval to market it abroad, with plans to launch third-phase clinical trials in the US and Europe in early 2020," the source added.
According to the World Health Organization, Alzheimer's disease, which begins with memory loss and develops into serious brain damage, causes 60% to 70% of cases of dementia reported worldwide. Dementia is estimated to affect about 50 million people worldwide, including 9.5 million people in mainland China, Hong Kong, and Taiwan.
Follow NEWS.am Medicine on Facebook and Twitter
- Video
- Event calendar
- Children’s Hospital Los Angeles and International Center of Professional Development Allergy/Immunology Conference
- First Armenian-German Conference entitled “Heart Failure Spring School”
- Allogeneic bone marrow transplant in case of hematological malignancy performed in Armenia for first time
All materials
- Archive
- Most read
month
week
day
- JAMA Oncology: Urine test can help rule out high-grade prostate cancer with almost 100% accuracy, study shows 1294
- Scientists grow human mini-lungs in lab 1174
- Next pandemic likely to be triggered by flu - scientists 943
- Scientists found baked goods and lack of sleep to be more dangerous than alcohol 916
- 342 cases of measles recorded in Armenia so far in 2024 868
- Scientists develop new method to safely stimulate immune cells to fight cancer 783
- Blood test can determine who is at risk of developing multiple sclerosis - scientists 781
- Cognitively stimulating jobs in midlife could lower dementia risk in old age, study finds 781
- BrainStimulation: electrical brain stimulation alleviates anxiety and depression in the elderly 721
- Ketamine may help with postpartum depression 650
- Unhealthy amount of sugar found in baby food products of a well-known brand 647
- Appetite: Scientists found out the secret to the appeal of large portions of fast food 644
- Air pollution puts health of more than 1.6 billion workers globally at risk 643
- Scientists test new approach to fighting viruses 629
- Zombie deer disease possibly linked to hunters’ deaths 536
- Find us on Facebook
- Poll